Hypochlorous acid underwear disinfectant | Sterilization 99.999%
| Name |
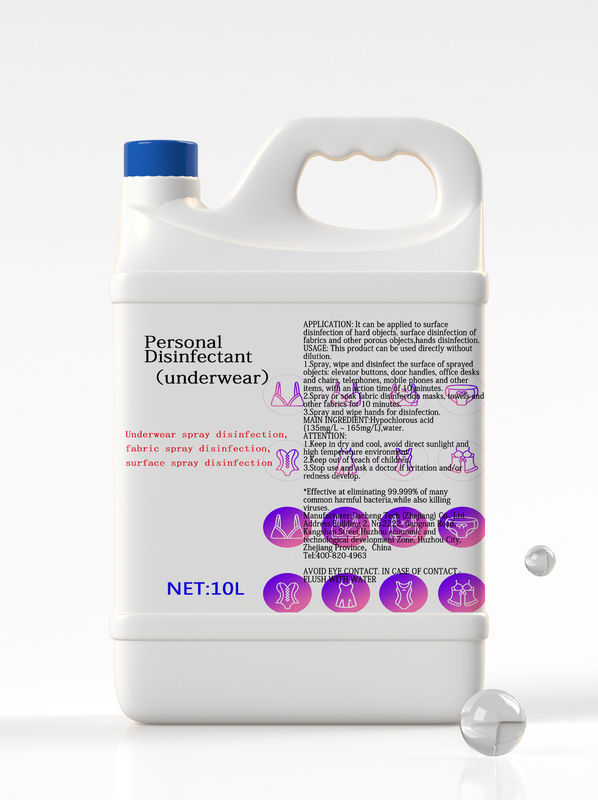
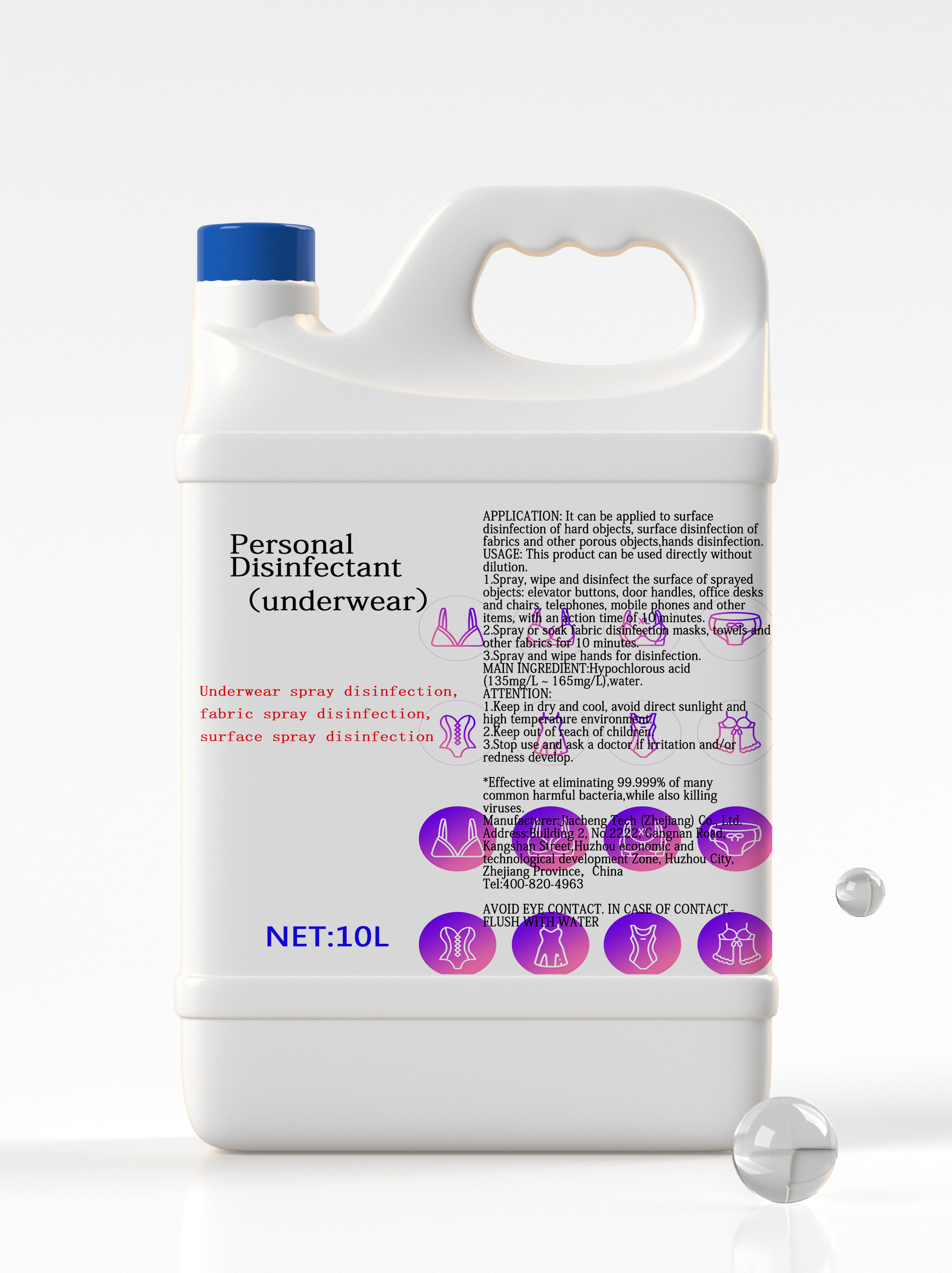
Personal Disinfectant(underwear) |
| Volume |
10L |
| Place of Origin |
China |
| component |
Hypochlorous acid 0.015%,Other ingredients 99.985%,Total 100.000% |
| Certification |
FDA,MSDS,CE,REACH |
| OEM |
MOQ 10000 pcs |
| Payment |
T/T |
| Delivery Methods |
DHL/FEDEX/TNT, by sea, by courier, by air. decided by clients |
| Delivery Time |
7 working days after payment |
| Supply Ability |
100000 Pieces Per Day |
| Instructions for use |
Underwear spray disinfection, fabric spray disinfection, surface spray disinfection |
| Features |
It can kill intestinal pathogenic bacteria, pyogenic cocci, pathogenic yeasts, and common bacteria in hospital infections. |
What is Hypochlorous Acid (HCLO)?
- Hypochlorite water disinfectant is a kind of small molecule compound, and its effective component is hypochlorous acid (HClO).
- Slightly acidic hypochlorite disinfectant refers to the pH value controlled between 5.0-6.5.
- Hypochlorite is one of the most effective bactericides produced by phagocytizing neutrophils in white blood cells.
- The micro acid hypochlorite produced by artificial technology is widely used for disinfection of indoor air, surface of objects, water, human skin and mucous membrane.
Hypochlorous acid (HOCL) is the simplest disinfectant with inside the chlorine own circle of relatives to be had in dilute solution. It is recommended that HOCL is eighty to one hundred twenty instances extra efficacious than sodium hypochlorite.

| PRODUCT |
Hypochlorous acid |
ALCOHOL |
BLEACH |
SANITIZER |
| Main Ingredient |
HCIO |
ALCOHOL |
NaCIO |
PCMX |
| Kills other bacteria |
Yes, it does |
No, it does not |
No, it does not |
No, it does not |
| Broad spectrum |
Yes, it does |
No, it does not |
No, it does not |
No, it does not |
| Disinfection rate from bacteria |
Yes, it does |
Yes, it does |
Yes, it does |
Yes, it does |
| No residue |
Yes, it does |
No, it does not |
No, it does not |
No, it does not |
| Could be used for air disinfection |
Yes, it does |
No, it does not |
No, it does not |
No, it does not |
| Non-irritation to skin |
Yes, it does |
No, it does not |
No, it does not |
No, it does not |
| Non-irritation to eyes and mouth |
Yes, it does |
No, it does not |
No, it does not |
No, it does not |
| Non Flammable |
Yes, it does |
No, it does not |
Yes, it does |
Yes, it does |
| Help to finish smell |
Yes, it does |
No, it does not |
No, it does not |
No, it does not |
Quaternary Ammonium (Quats) VS Hypochlorous Acid
HOCL has always considered better than many other chemicals of its type. For example, Quats while purifying food contact surfaces since Quats are not a better option in terms of safety on food. Prior to cleaning with chemical like Quats, all food must be eliminated from the region before cleaning can happen. This isn't needed when you use HOCL. Hypochlorous Acid can be used almost all of the day for the food and it won't decay the food.
Ozone VS Hypochlorous Acid
Hypochlorous acid has always considered better than many other chemicals of its type. For example Ozone, Ozone is a gas and it is unstable in mixtures due to that reason it can't be used to sterilize contact surfaces. Ozone is anyway used for food sterilization anyway it should again and again be recovered as it is constantly evaporates into the air.
Being that ozone is a bad option for the lungs and respiratory system, ozone is restricted in the focuses that can be used. Due to that cause it is a restriction that can be used for the microbial microorganisms(microbial pathogens). Hypochlorous acid does cause any kind of irritation and is likewise stable in mixture. Therefore, HOCL can be used at high fixations (60 ppm) for food disinfection the need of a post-wash.
Hypochlorous acid has obvious deodorizing effect
Once a Japanese researcher used a sample bag to collect the odor, then added tap water and hypochlorous acid to the sample for a control test. After 30 times of violent vibration, the FF-2020 gas recognition device was used for odor analysis. The results are as follows:
| Substance name |
Hypochlorous acid |
Odor index equivalent |
| |
Available chlorine concentration
ppm
|
Tap water |
Hypochlorous acid |
| Hydrogen sulfide |
18.4 |
30 |
22 |
| Dimethyl disulfide |
18.2 |
25.3 |
0 |
| ammonia |
18.4 |
12.6 |
5 |
| Trimethylamine |
18.1 |
22 |
13.5 |
| Ethanol |
18.9 |
19 |
17 |
| Butanol |
18.4 |
26 |
6 |
| Ethyl acetate |
18.8 |
19 |
19 |
| Propionic acid |
18.4 |
23.6 |
0 |
| Butyric acid |
20.1 |
31 |
22 |
| N-valeric acid |
18.9 |
29 |
23 |
| Isovaleric acid |
20.2 |
31 |
22 |
It can be seen from the above that the low concentration of hypochlorous acid removes the spoilage odor of vegetables (methyl mercaptan, dimethyl sulfuric acid, dimethyl disulfide), toilet odor (ammonia), livestock and poultry breeding odor, pungent sour odor (propionic acid) ) The ability to reach more than 99.99%, to deodorize rotten egg smell (hydrogen sulfide), smelly fish smell (trimethylamine), sweat smell (n-butyric acid), footwear smell (n-valeric acid, isovaleric acid) The capacity can also reach about 60%. If the effective concentration of hypochlorous acid is appropriately increased, its deodorizing effect will be doubled.
Therefore, when faced with these odors, hypochlorous acid can chemically react with these odors to neutralize and decompose these odors to achieve a good deodorizing effect!
 Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!  Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!